 |
48 (1) (1996), pp. 14-21. JOM is a publication of The Minerals, Metals & Materials Society |
|---|
 |
48 (1) (1996), pp. 14-21. JOM is a publication of The Minerals, Metals & Materials Society |
|---|
"All nonferrous metallurgy will be benefited by the use of cheap oxygen . . . the application of oxygen will revolutionize the art of smelting and it will probably change the whole operation and equipment."
So wrote F.W. Davis of the U.S. Bureau of Mines almost 75 years ago.1 He cautioned that the existing cost of oxygen was much too high for metallurgical processes, but he forthrightly stated that "the oxygen industry is now able to make plants for supplying large quantities of oxygen to metallurgical industries at low cost." His committee's convincing Report of Investigations—primarily concerned with steelmaking—was aimed at major decreases in metal production cost. There was no consideration of environmental impact—this subject was not considered important in 1923, and the opinion would not change for decades. As pioneer environmentalist, scientist Rachel Carson warned, "A grim specter has crept upon us almost unnoticed."2 Today, engineers must heed the bell that she first tolled.3 ,4
Davis' prescient words fell on deaf ears throughout the world's nonferrous industry. Ultimately, however, they sounded in Inco's Copper Cliff Research Laboratory in 1941 and were amplified by the pertinent 1936 study of Telfer Norman5—but there was a war to win. When the veterans returned, the "innocently beloved proud plumes of heavy industry" that were billowing out of the Copper Cliff Smelter stacks were recognized for what they were. Most notably, their sulfur content was wounding Ontario and Quebec forest lands. Simultaneously, the paper-making sulfite pulp mills were importing large quantities of elemental sulfur, and the smelter was importing large quantities of coal.
Economic application of oxygen pyrometallurgy would permit replacement of this sulfur by liquid sulfur dioxide, with simultaneous replacement of the coal by low-cost tonnage oxygen, produced using low-cost hydroelectric power. Reaching this grail would achieve not only a significant decrease in metal production cost, but it would also enable an significant decrease in environmental degradation, a goal that would be increasingly stressed by government.
Inco's resulting half century of progress in developing oxygen pyrometallurgy (Figure 1) is a paradigm of the long-term teamwork that is necessary to attain such an operational shift—laboratory theoreticians, hot-metal operators, and management all working together with mutual respect.
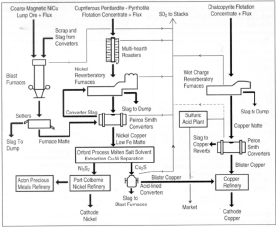
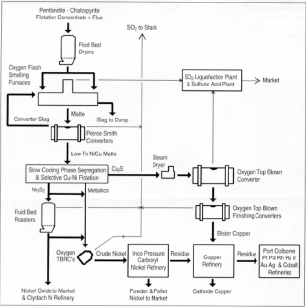
Figure 1b. Flowsheets of nickel-copper extraction at Copper Cliff—1995.
Assistance was, therefore, sought from sound, though relatively inexperienced, sources, and the low bidder was accepted. It was the parent of Canadian Liquid Air—Air Liquide of Paris, which was then a builder of small oxygen plants; it is now the builder of the largest oxygen plants in the world. The fearful cost barrier was hurdled by the determining contributions of their brilliant chief engineer: Maurice Gobert. A worthy bid was also submitted by enterprising Leonard Pool, founder of Air Products, the supplier of reliable mobile dwarf oxygen generators for U.S. Army Engineers in World War II; Air Products is now a giant tonnage oxygen supplier.
In January 1948, Copper Cliff managers Roy Gordon (plant) and Paul Queneau (R&D) submitted their joint decision to top management:
"In view of the economic superiority and metallurgical potentialities of the new process it is recommended that a (first step) 400 t.p.d. copper concentrate flash smelting unit and a 300 t.p.d. oxygen plant be installed at Copper Cliff. . . . There would be periods of large surplus oxygen production . . . such oxygen could be consumed to advantage in the Smelter reverberatory furnaces for air enrichment so as to decrease coal consumption."
In those early post-war years, there were very long delays in equipment delivery, particularly of special machinery for the oxygen plant. A 500 t/d copper concentrate oxygen flash smelting furnace—supplied with low-cost oxygen by a 300 t/d plant—finally went on-stream four years later: January 2, 1952.6
This energy efficient and environmentally friendly reactor led the world of pyrometallurgy—including the oxygen steel converter—in its direct, massive-scale, use of oxygen. In the spring of 1948, convincing experiments in Gerlafingen, Switzerland, inspired by Robert Durrer, demonstrated oxygen top-blowing of blast-furnace hot metal into steel. In November 1952, VOEST's oxygen steelmaking Linz/Donawitz converter plant went on-stream in Austria.7
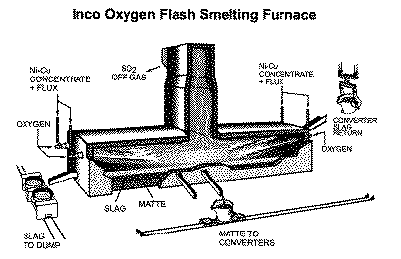
A pilot-plant furnace was then designed and built for the continuous smelting of dry sulfide flotation concentrates. A design principle was "keep it simple stupid," so both furnace and burner were basically adaptations of the neighboring reverberatory furnaces—with mineral concentrate and oxygen substituted for coal and air. Another difference from conventional practice was cyclic reduction drenching of furnace slag with molten FeS from flash-smelted pyrrhotite concentrate prior to tapping, which decreased slag oxygen potential and CuNi loss. This practice would have been improved by the inclusion of some coal with the pyrrhotite, but this "obvious" action was not obvious at the time. Oxygen was supplied at 95% O2 by a Canadian Liquid Air 5 t/d oxygen plant, and furnace off-gas was treated in a Canadian Industries Limited sulfur dioxide liquefaction pilot plant.
Pilot-plant operations started in January 1947 and were terminated late in 1948 with full success at 25 t/d of concentrate. Furnace dimensions had to be increased four times—mainly due to refractory erosion by flying molten particulates—before the final size was attained. Matte grades of up to 75% CuNi were produced, with pyrrhotite-cleaned slags analyzing up to 0.9% CuNi. Testing of the liquid SO2 product for sulfite pulp production by Abitibi Paper Company indicated its superiority to SO2 produced by sulfur burning.
The day after New Year's Day, 1952, a pioneering commercial oxygen reactor (Figure 2) flashed into life. In accordance with prior planning, the new plant superintendent—plant coinventor Charles Young—was the former pilot-plant superintendent; before that, he had been a laboratory engineer involved in the initial R&D. The able, invaluably supportive, up-from-the-ranks smelter manager, Duncan Finlayson, had originally been contemptuous of the "black box" furnace concept: "I can't stick my head in it—I don't like it!" His understandable skepticism was overcome by early pilot-plant exposure and by the effectiveness of the well-maintained instrumentation employed, which allowed the operator to monitor and control the key furnace variables. After the usual birth pains, the daily furnace charge averaged 500 tonnes of 28% Cu chalcopyrite concentrate, 100 tonnes of pyrrhotite concentrate, and 90 tonnes of sand flux. The matte produced analyzed 45% CuNi, the slag was 0.75% CuNi, and the off-gas was 75% SO2. The last—relatively small in volume—was condensed in a 300 t/d liquid SO2 plant, which was a decimal order of magnitude larger than any other in the world.8
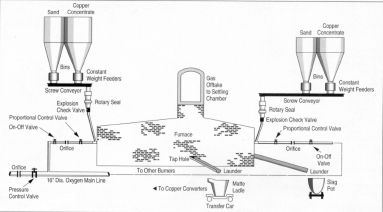
Having well served its educational function, the innovative furnace was replaced by a rewardingly profit-making 1,000 t/d unit two years later. The critically vital, Inco-owned and operated Air Liquide tonnage oxygen plant delivered admirably—producing 95% O2 gas at a total cost of $4/t and using 0.4¢/kWh power. Low-cost production of pipeline tonnage oxygen was proven! Pyrometallurgy was reborn! The Inco staff technical paper describing this achievement,6 and the five names in alphabetical order on the covering patent9 complied with the aphorism: "Share credit, share success." Now, Inco owns and operates Air Liquide oxygen plants with a total capacity of 1,800 t/d to feed a variety of Copper Cliff furnaces. 10
There were 19 air-blown 4.0 m X 10.7 m Peirce-Smith converters in the Copper Cliff main converter aisle, treating liquid reverberatory furnace matte. Oxygen utilization efficiency, in its exothermic reaction with iron and sulfur, was close to 100%. However, half of the total heat developed suffered the nitrogen curse, so it was lost. Several of these converters blew flash-furnace matte to blister copper. If blower air was enriched with oxygen (e.g., to 33% O2, thereby changing N2/O2 volume ratios from 4:1 to 2:1), the usual heat balance in the converter would be much improved. In addition to the increase in the conversion rate and decrease in gas volume, cold charge could be smelted. The limiting factor would be excessive impact on tuyere and refractory life. Converter trial operations, in which the oxygen content of blower air was varied in the 25-35% range, were launched in 1958. These indicated a 30% O2 content to be optimal. The additional useful reaction heat was employed to melt large quantities of scrap and concentrates (e.g., the 73% copper filter cake produced by the matte flotation copper-nickel separation plant). The 30% O2 level of enrichment was then systematically extended to all 19 converters.
On the basis of Inco's experience with tonnage oxygen generation and utilization, one of the authors was able to write the following in 1960:
"The pyrometallurgist will gain further benefits from the advent of low-cost oxygen. The dead hands of nitrogen have been lifted from oxidation reactions which utilize the oxygen in air. The nonferrous metal industry is on the threshold of understanding in this connection. As one example, oxygen enrichment of combustion air will give new life to otherwise obsolescent or obsolete conventional furnaces. Greatly improved reverberatory and rotary furnace design will be employed for utilization of tonnage oxygen in continuous autogenous smelting and converting. The tuyereless, top-blown oxygen steel converter will invade and conquer the smelters and refineries of the nonferrous industry. Decrease in nitrogen dilution of sulfurous smelter gases will permit increased sulfur fixation and result in decreased atmospheric pollution."12
This prediction has proven accurate.13-17 Nevertheless, low-cost oxygen—the immense value of which was demonstrated on a commercial scale in 1952—continued to be greatly underemployed for decades. 18,19
However, 1941 Copper Cliff Laboratory studies had indicated that conversion was possible, given sufficiently high bath temperature and oxygen potential. Hence, the high-temperature, broad-range oxygen potential and excellent mixing capabilities of the turbulent bath, characteristic of a post-war top-blown rotary converter (TBRC) steelmaking process, appeared extremely attractive. The vessel employed provided efficient and effective gas-liquid-solid contact throughout the bath—with concomitant extraordinarily extensive control of temperature and oxygen potential. It enhanced heat transfer, increased the overall rate of the chemical reactions, minimized composition gradients within each phase, and significantly reduced diffusion barriers.
Oxygen metalmaking by tonnage nickel matte experiments in a TBRC were proposed and opposed in heated debates within Inco. Conventional wisdom said such experiments would fail and perhaps kill: the converter would produce nickel oxide instead of metal, and the nickel sulfide (melting point— 788°C), at 1,650°C, would cut through the rapidly rotating refractory lining like a knife through butter.
In 1958, Paul Queneau and John Feick, Copper Cliff Peirce-Smith converter superintendent, supported by John Thompson, Inco's chemical engineer chief executive officer, explored direct nickel sulfide conversion to oxygen crude nickel in a three tonne KALling converter at DOmnaverts Steel Works (KALDO) in Sweden. The experiment was immediately successful. This victory in novel nickel making having been achieved, opportunities in TBRC oxygen coppermaking, fire refining, and beyond were revealed and successfully pursued by Inco in a seven tonne TBRC at Port Colborne.20,21 It all seemed so obvious after the breakthrough.
In 1971, two 50 tonne TBRCs were commissioned at Copper Cliff (Figure 3) as the first stage in the transformation of complex metal sulfide intermediates to 99.98% pure nickel by the Inco Pressure Carbonyl Process. Today, the operation of these converters is routine, having produced a million tonnes of oxygen crude nickel to date.22

Commissioning and operation of the TBRCs completed development of an oxygen culture at Copper Cliff. Management and technical staff understood the advantages oxygen technology offered, and operators and maintenance personnel knew how to work with oxygen as a useful ally. This culture, coupled with continuing active research and development, enabled commercialization of new oxygen technologies as Inco responded to the changing economic and environmental challenges of the seventies and eighties.
These second-generation Inco oxygen flash furnaces (Figure 4) are larger than the original furnaces, employ greater amounts of water cooling, and incorporate modern gas cleaning systems that are extremely compact, as allowed by the low-volume exhaust gas of tonnage oxygen smelting. Cleaned gas feeds a double-contact acid plant and the original liquid SO2 plant. 10,26 Furnace feed is a bulk copper/nickel concentrate. Petroleum coke and natural gas are added to provide supplemental heat and to allow return of converter slag and smelting of reverted material. Table I compares the original and new furnaces.

Development of a novel oxygen-based flash-converting process gave a short-term, low-capital improvement.28, 29 Due to the rapid kinetics of oxygen reactions, the smelter was able to use a surplus Peirce-Smith converter shell as the vessel. The in-house development of a suitable feed system and unique oxygen flash gun that could simultaneously fire natural gas and filter cake completed the process. This process, the first commercial application of flash copper converting, started in 1985 and operated for eight years, treating 8% moisture filter cake at rates of 250 t/d to 300 t/d. More than 300,000 tonnes of molten semiblister assaying 2-3% sulfur were produced.
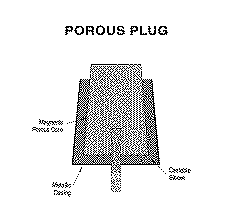
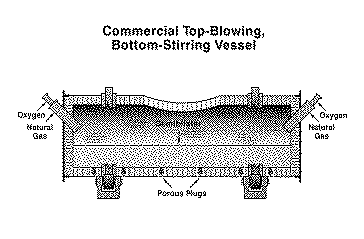
Full scale tests began in 1989 using a Peirce Smith converter shell equipped with two porous plugs and an oxygen lance. The combined blowing approach yielded oxygen efficiencies of 85% during blister finishing, although the subsonic open pipe lance was mounted 1.8-3.7 m from the bath and blew gently to minimize splashing. By using oxygen, the converter consumed scrap at a rate of 20% of the semiblister charge (2-3% sulfur). The porous plugs performed well in copper service, and elimination of tuyeres minimized fugitive emission generation when the converter rolled into and out of stack. Finishing blister by oxygen top blowing/nitrogen bottom stirring was incorporated into the new flowsheet of the Copper Cliff Smelter in 1990.28 To conserve capital, the process was implemented in existing Peirce-Smith converter shells (Figure 6). Commercial operation began in November 1993. *
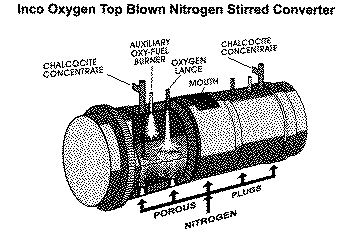
Plant tests showed that tuyere injection of chalcocite accompanied by oxygen top blowing/nitrogen stirring was effective, and commercial operation commenced in 1993.36 The reactor vessel is a 18 m long, 4.5 m diameter cylinder with oxygen lances mounted on each endwall. Each of two blow-tank conveying systems is connected to a single tuyere and injects chalcocite at a rate of 25 t/h. Dusting rate is about 1%, and oxygen efficiency is in the 90% range.
Pilot-plant studies in 1994 showed the feasibility of combining top blowing/bottom stirring with a simplified feeding technique. Full-scale tests began in August 1995. The application of nitrogen stirring through porous plugs has been extended a further step. Feeding is accomplished by gravity introduction of dry, nonagglomerated concentrate (90% -44 µm) through a water-cooled pipe onto the "eye" created by the nitrogen. 37 Supplemental heat is provided in the area to promote melting. Feeding zones and converting zones are separated so that gas velocity around the feed stream is minimal (Figure 7). As a result, a dusting rate of 1.5-2% is achieved. Full-scale development continues. As demonstrated in the pilot-plant work, this simple approach can be useful for other continuous converting applications.
The vacuum swing adsorption process has found application for oxygen requirements of less than 100 t/d at purities of 90%. In this technique, nitrogen is removed in two molecular sieve adsorption trains connected in parallel and operating in sequence. While incoming air is purified in one train, the other train is regenerated by pressure reversal. 40 Such an installation can be used to supply oxygen to a relatively small user or to top-up a large cryogenic plant that cannot meet ever-increasing smelter demands.
Algorithms that account for both mass and heat effects in autogenous or semiautogenous reactors require comprehensive information about input and output streams. Using distributed control systems, the solid-feed rate, typically controlled with impact-type meters, can be systematically calibrated with more accurate weight loss readings from dry feed bins. These bins are subject to both filling and emptying cycles; thus, direct use of weight loss is not possible.
Recent developments in analytical techniques promise a revolution in the determination of solids composition. Prompt gamma neutron activation spectroscopy is employed in power and cement plants to provide on-line analysis.43 The solids are irradiated with neutrons and, in turn, emit gamma rays characteristic of the nuclei present and independent of matrix effects. Analysis of the spectra involves significant data processing. However, once set up, the technique can be used for continuous measurements over a moving belt. Alternatively, the technique may provide quick chemical assays with simplified sample preparation steps amenable to the shop floor.
Monitoring of pyrometallurgical processes is seriously hampered by the vulnerability of sensors to high temperatures. Moreover, liquid and solid particulates in the reactor atmosphere cause corrosion and erosion. Direct temperature measurement by insertion of thermocouples into the reactor freeboard is often impractical because they burn or short out. The development of two-wavelength pyrometers has improved temperature measurement, but even these pyrometers can be affected by the atmosphere. Thus, temperature in many pyrometallurgical reactors is currently determined by manual immersions during skimming and tapping. An interesting approach, developed by Noranda, is temperature measurement through tuyeres.44 A retractable periscope mounted on the back of a tuyere transmits light via a fiber-optic cable to a two-wavelength pyrometer located remotely from the reactor.
Accurately measuring matte and slag levels in a closed reactor is difficult. The widely used technique of bar immersion is distinctly limited with respect to both accuracy and applicability. Determination of reactor weight,45 gamma radiation, lasers, and microwaves can be used to measure levels of molten systems.
Gamma gauges for remote determination of bath levels have found application in the glass industry.46 The apparatus is mounted in the narrow forehearth area of the furnace and comprises a transmitter located on one side and a receiver on the other. Application of the technique to nonferrous applications (e.g., to indicate slag, matte, and metal levels) has so far been limited due to large reactor widths.47
Laser-based systems have found application in casting operations. In the aluminum industry, lasers measure liquid levels in furnaces and also the rate of mold fillage.48 Similar applications apply to cast iron operations.49 The application of lasers in nonferrous reactors may be limited by the presence of dust and fume.
Electromagnetic microwaves hold promise for determining bath levels in continuous oxygen pyrometallurgy. Microwaves are relatively insensitive to smoke and dust and are not affected by high temperatures or temperature gradients as are ultrasonics. In the steel industry, radar has been employed to measure metal level in basic oxygen furnaces and torpedo cars. 50,51 Microwaves can also quantify the rate of rise during bottom teeming.52 A method of measuring slag thickness during casting operations has been identified.53
Monitoring the progress of a steel converter with disposable oxygen probes based on stabilized zirconia has long been an accepted part of the process. Commercial applications in nonferrous systems are more limited. Today, highly reliable probes for measuring bath oxygen potential in copper converters and anode furnaces are available,54 and their use will increase. However, the much desired continuously operating oxygen probe remains elusive due to the sensitivity of the electrolyte. Other solid electrolyte systems are sensitive to CO2, SOx, and NOx and may find commercial application in pyrometallurgy.55 Still other potential methods of monitoring reactor conditions include optical spectroscopy56 and continuous analysis of internal and exhaust reactor gases (e.g., O2, CO, CO2, H2, SO2) by employing in-situ probes or sample withdrawals.
"The overall cost advantages which accrue from continuity—not least in respect to environmental conservation—are manifest. . . .There is no reason why hydrocarbon-shielded oxygen jets cannot be advantageously employed for continuous subsurface-blowing in nonferrous converting practice."60 In 1974 the Q-S continuous oxygen converter was publicized throughout the United States and was illustrated on the cover of JOM. The inventors believed it would "prove to be a contribution to maximum economic utilization of the nation's mineral heritage, with due regard to conservation of natural resources—including the environment." 61,62 Two decades later, commercial QSL (Queneau-Schuhman-Lurgi) oxygen converters are continuously making metal directly from mineral feed. A dream is finally a reality!63-66
There are, of course, other dreams being pursued. For example, industry needs to fully harness the energy released by the oxidation of SO2 to SO3 during acid production. This energy could often produce sufficient 40 ats steam, for power generation, to supply all or most of the amount required for oxygen production. We also need to improve our ability to control the process parameters that characterize the ideal pyrometallurgical reactors of the future. These will rapidly and continuously convert mineral sulfide concentrate and appropriate recycled materials to acceptable quality metal, clean slag, and sulfur dioxide-rich gas by fully utilizing the concentrate's natural fuel content in closed, fugitive emission-free reactors. The chemical and steel industries are making great strides in process monitoring (e.g., tomography), and the nonferrous industry must also follow their lead. 67,68
Oxygen pyrometallurgy has revolutionized the industry. The changes it has wrought can be compared with developments of the turn-of-the-century decades (e.g., multihearth roasters, Dwight-Lloyd sintering machines, huge reverberatory and open hearth furnaces, by-product coke ovens, and Peirce-Smith converters). Today, oxygen usage is ubiquitous and addictive. Substituting oxygen for air vastly increases process productivity and cleanliness. Revolutionary sparks were ignited at Copper Cliff and Gerlafingen half a century ago. However, until the winds of energy conservation and environmental protection blew compellingly, the fires were confined. Now the fires burn briskly around the world—the future of pyrometallurgy is bright!69-71
*Development of porous ceramic plugs began in 1947 and was led by Steven Spire and Robert Lee of Canadian Liquid Air in conjunction with the Canadian Bureau of Mines.3 In the 1970's, porous plug use became widespread in the steel industry. However, their use in nonferrous pyrometallurgy has been limited, and the usage described here represents its first commercial application in copper smelting.
For more information, contact P.E. Queneau, Thayer School of Engineering, Dartmouth College, 8000 Cummings Hall, Hanover, New Hampshire 03755-8000; fax (603) 646-3856.
Direct questions about this or any other JOM page to jom@tms.org.
| Search | TMS Document Center | Subscriptions | Other Hypertext Articles | JOM | TMS OnLine |
|---|