Overview
Steel vs. Plastics: The Competition for Light-Vehicle Fuel Tanks
Peter J. Alvarado
As weight and cost savings drive changes in performance criteria for
automotive materials, original equipment manufacturers (OEMs) are taking a
harder look at the historically terne-coated steel used for gas tanks. This
article compares steel and plastic for gas tank uses according to performance
attributes and a competitive analysis. Legislation issues and current OEM
activity are included.
Historically, terne-coated steel (an 8% tin-lead coating) has been the
mainstay for automotive gas tanks; however, several issues are changing the
performance criteria that must be met and, thus, threaten the application of
steel products. The drivers for a material change are legislation, increased
required part life to ten years/241,350 kilometers, permeability, weight,
packaging, safety, and cost. In this article, the performance attributes of the
plastic and steel alternatives are reviewed from an original equipment
manufacturer (OEM) perspective in the critical areas of manufacturability,
cost, design, weight, safety, corrosion, and recyclability.
A comparative analysis of the various plastic and steel alternatives indicates
that steel remains a cost-effective material that meets all of the required
performance criteria. A more specific cost comparison of the new plastic tanks
(i.e., multilayer or barrier coated) with the new steel tanks is still
required. Many of the drivers such as lead reduction, clean fuels,
permeability, and weight are a direct result of legislative and regulatory
pressures described in the Issues sidebar.
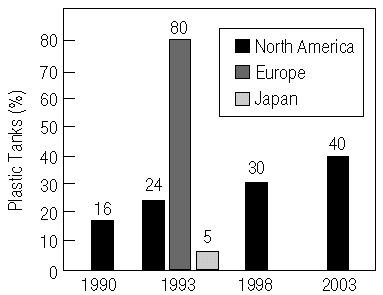
Figure 1. The 1998 and 2003 projections of plastics usage in fuel tanks, based on the Delphi VII report. |
Since the mid-1980s, automakers have been displacing coated-steel fuel tanks
with plastic ones. During the 1993 model year, approximately 2.7-3 million cars
and trucks built in North America used nonmetallic tanks. This represents
22-25% of the market, compared to 16% in 1990. By comparison, the European
market uses 70-90% plastic tanks, and the Japanese market uses 5% plastic
tanks4 (Figure 1).
Although plastic-tank applications have experienced some reversals as a result
of the stricter permeation standards, some experts believe their usage will
gain momentum by the end of the decade as new plastics technology is converted
to commercially feasible operations. The Delphi VII report by the University of
Michigan indicates that experts predict plastic tanks will capture 28% of the
North American market by the end of 1996 and up to 50% of the market by
2000.5 However, this projection needs to be tempered with the higher
manufacturing cost and recyclability issues of the multilayer plastic tanks
that will be required to meet the stricter permeation standards.
This projection is considered as the worst-case scenario for steel if the
industry fails to provide the OEMs with a cost-effective steel alternative that
meets all of the performance criteria. Table I indicates the production volume
of vehicles built in North America;6 Table II shows the estimated
number of plastic gas tank units (according to Delphi VII projections) and
their impact on steel shipments.
As of 1993, the steel industry lost the opportunity to ship about 34,473
tonnes of steel as a result of plastics gains in gas-tank applications. In the
worst-case scenario, continued acceptance of plastic gas tanks will increase
steel's loss to 71,667 tonnes in 2000—an additional 43,544 tonnes. This means
that annual shipment of 125,191 tonnes of steel will be reduced by better than
a third to a total of about 81,646 tonnes per year.
| Table I. Actual and Projected Car and Light Truck Production in North America (in thousands)6 |
|---|
| 1992 | 1993 | 1994 | 1995 | 1996 | 1997 | 1998 | 1999 | 2000 |
|---|
| Cars | 7,474 | 8,141 | 9,050 | 9,775 | 9,960 | 10,125 | 10,280 | 10,358 | 10,435 |
| Light Trucks | 5,033 | 5,709 | 6,535 | 6,749 | 6,923 | 7,008 | 7,088 | 7,103 | 7,117 |
| Total | 12,507 | 13,850 | 15,585 | 16,524 | 16,883 | 17,133 | 17,368 | 17,461 | 17,552 |
| Table II. Projected Plastic Gas Tank Share Based on the Delphi VII Scenario |
|---|
| Share (%) | Steel No. of Tanks | Steel Lost (tonnes) | Change Incre. (tonnes) | Change Cum. (tonnes) |
|---|
| 20 | 2,501,000 | 28,365 | - | -
|
| 22 | 3,047,000 | 34,552 | 6,187 | 6,187
|
| 24 | 3,740,000 | 42,414 | 7,862 | 14,049
|
| 26 | 4,296,000 | 48,717 | 6,303 | 20,352
|
| 28 | 4,727,000 | 53,605 | 4,888 | 25,240
|
| 30 | 5,140,000 | 58,284 | 4,679 | 29,919
|
| 32 | 5,558,000 | 63,022 | 4,738 | 34,657
|
| 34 | 5,937,000 | 67,319 | 4,297 | 38,954
|
| 36 | 6,319,000 | 71,651 | 4,332 | 43,286
|
| Incre.—incremental; Cum.—cumulative. |
High-density polyethylene (HDPE) has been the resin of choice for plastic gas
tanks, and production capacity has been on the increase. Kautex of Canada built
a new plant in Avilla, Indiana, to meet anticipated increases in demand for
plastic automotive fuel tanks. Production was scheduled for 400,000 tanks in
1994 and eventually will be boosted to between 600,000 and 700,000 units
per year.7 These plastic tanks are currently being used on
Chrysler's Jeep Cherokee and T300 trucks.
Belgium-based Solvay is the exclusive supplier of plastic fuel tanks to
General Motors' Saturn Division. Solvay has also expanded their Canadian
subsidiary in Blenheim, Ontario, and installed two new blow-molding machines to
make HDPE tanks for the Chrysler's LH series and Viper sports car. Chrysler
expects to sell 300,000 LH vehicles, all with HDPE tanks that offer more volume
capacity than steel tanks. Monolayer-HDPE tanks offer long-term structural
integrity but will not meet future permeation requirements. Chrysler started to
switch in 1995 to multilayered HDPE to meet the more stringent SHED test.
The emergence of new technologies has enabled the increase of plastic gas
tanks. These new technologies can be grouped into either multilayer or barrier
types.
Some manufacturers see multilayer tank technology as the answer to
stricter emission standards. Ford uses six-layer fuel tanks made of HDPE and,
at one point, considered a $110 million investment in machinery and equipment
to produce the tanks (which would have been the first commercial use of
coextruded tanks). However, Ford decided to increase steel-tank stamping
capacity at Dearborn; the Explorer and the new F-150 truck (PN96) gas tanks
have been switched back from plastic to zinc-nickel coated steel.8
Kautex is supplying Ford's facility in Milan, Michigan, with this six-layer
technology. The tanks are designed to meet California's stricter evaporative
fuel standards and consist of an inner layer of HDPE joined by an adhesive
layer and barrier layer of polyamide or ethylene-vinyl alcohol copolymer. An
additional adhesive layer is joined by a layer of "regrind" and an outer layer
of HDPE.
Walbro Automotive Corporation began commercial production of multilayer
plastic fuel tanks for 1995 models. Annual production of these multilayer tanks
is expected to reach 500,000 units by 1998. Their tank uses a barrier layer of
ethylene-vinyl alcohol (EVOH) that is sandwiched between two layers of
HDPE.9
| LEGISLATION AND REGULATORY ISSUES |
| A number of federal and state legislative and regulatory actions have been initiated to promote the use of cleaner automotive fuels such as reformulated
gasoline, methanol, ethanol, and natural gas. In 1988, the U.S. Department of
Transportation introduced a Clean Fuels Grant Program for local transit
authorities. The Alternative Motor Fuels Act of the same year established
production incentives and a federal purchase plan for alternative fuel
vehicles. Later, the Clean Air Act Amendments of 1990 established fleet
mandates to reduce emissions from the primary source of air pollution (i.e.,
vehicles). Two years later, the Energy Policy Act of 1992 encouraged the use of
alternative-fuel vehicles or flexible-fuel vehicles (FFVs) to reduce oil
imports and increase the use of U.S. sourced and renewable fuels. In addition,
future increases in Corporate Average Fuel Economy (CAFE) will add pressure to lighten every vehicle component, including gas tanks. These issues and
stricter U.S. Environmental Protection Agency (EPA) fuel-permeation standards
are challenging the designs and materials used in vehicle fuel tanks.
Flexible-Fuel Vehicles
The automotive industry has been pursuing ways to meet the legislative and
regulatory pressures to reduce emissions in the form of FFVs capable of running
with methanol/gasoline mixtures. However, testing has verified that neither
bare nor painted terne will resist the corrosion effects of alcohol (methanol
or ethanol)-based fuels for the expected life of the vehicle—241,350 km or ten
years.
The actual methanol fuel considered was M85 (85% methanol, 15% gasoline);
however, this fuel has recently lost popularity because, although it costs
about the same as gasoline, it offers only half the energy density by volume.
In the case of ethanol, the EPA and others estimate wholesale costs between
$1.50 and $2.10 per gallon.1
More recently, natural gas has pushed high methanol and ethanol fuels to a
very distant second and third in popularity. Natural gas is affordable—$0.70
worth provides the same energy of one gallon of gasoline. Additionally, it
produces no evaporative emissions and meets ultralow tailpipe
emissions.2 Furthermore, there is already a pipeline infrastructure
which, coupled to a low-cost compressor, would allow owners to refill at home.
However, compressed natural gas cylinders are bulky and heavy, thus reducing
space and range. Natural-gas vehicles, like electric vehicles and hybrids,
stress the need for weight reduction to achieve the same performance of
driveability and range as current automobiles. So do hydrogen-powered vehicles,
which represent another alternative that requires heavy tanks. The need for
lightening this type of vehicle also increases the possibility of alternative
materials substitution for steel parts in other components throughout the
vehicle.
Permeability/Evaporative Emissions
Three types of evaporative emissions are being studied: refueling vapor,
running (non-tailpipe) emissions, and permeation. Also, because of
environmental pressures, the materials used to contain the fuel and deliver it
to the engine are expected to be recyclable. The EPA currently uses the sealed
housing evaporative determination (SHED) test to measure pollution caused by
fuel evaporation. All major vehicle-manufacturing nations except Japan have
accepted SHED as the evaluation method of choice. The current SHED requirement
of 24 grams maximum of hydrocarbons in 24 hours has been tightened to the
more restrictive level of two grams per day.3 The material
implications for steel are tighter demands on the integrity of tank seams and
filler neck joint areas. Implications for plastics are the need to switch to
harder-to-recycle material systems and more expensive processes to meet the
stricter SHED test. |
Air Products and Chemicals of Allentown, Pennsylvania, has
commercialized a fluorine-based barrier technology that enables plastic fuel
tank manufacturers to meet more stringent emissions standards. The SHED tests
completed in 1992 on Airoguard plastic tanks produced by Kautex of Canada
indicated hydrocarbon permeation rates as low as 0.1 g/24 h—significantly
lower than rates for tanks using previously available barrier technology. The
performance of the Airoguard tanks compares with multilayer extrusion tanks
while maintaining the long-term structural integrity of monolayer
tanks.10
Solvay Automotive of Troy, Michigan, has developed technology to
keep HDPE tank emissions down to 0.2 g/24 h or less, even with
methanol-containing fuels.11 Using technology called
Solvay-optimized fluorination, Solvay equals or exceeds the permeation
performance of coextrusion with EVOH barrier resin. Solvay continues to add
multilayer capacity to meet Chrysler's requirements.
Aero Tec Laboratories of Ramsey, New Jersey, has developed a semiflexible
safety fuel tank made from an undisclosed olefin compound of polymers and
antidiffusion-barrier additives.12 The tank can be used for race
cars and military vehicles as well as general automotive aftermarkets.
North American auto manufacturers are currently supplied with tanks comprising
a steel substrate coated with either terne or zinc-nickel. In all, about
125,191 tonnes of steel substrate per year are applied to gas tanks.
Stainless steel tanks have been tested, and although effective in flexible
fuels, they are difficult to form without severe breakage occurring during
stamping. Also, stainless steel is expensive, with an estimated cost ratio to
terne steel exceeding 5:1.
The electrocoated zinc-nickel product is painted on both sides with an
aluminum-rich epoxy. Industry accelerated tests on the corrosion of painted
zinc-nickel confirm that it will meet a ten-year life in current fuels and
flex-fuels and resist external corrosion.
Testing the characteristics of painted galvanneal (zinc-iron alloy coated
steel) have found it effective for resisting corrosion on both the inside and
outside surfaces of the tank. General Motors has a product undergoing testing.
Hot-dipped tin has also been found to be effective for resisting all fuels,
but it does require a paint coating for exterior protection from road-induced
corrosion. This product welds faster than painted terne and has a better
potential for good solderability than painted galvanneal and zinc-nickel coated
steel substrates, permitting the attachment of fuel filler tubes and other
lines.
Terne plate holds a materials cost advantage over HDPE: $0.66-0.79/kg
vs. $0.86-1.08/kg.13 The cost of the material is not the only
driver; consideration includes the net cost of the fabricated tank and its
reliability within the total fuel system of the vehicle, including the tank,
filler tube, level control, baffles, the housing reservoir for the sending
unit, and other assorted tubing, fittings, and seals from the tank to the
engine. All of these components must function properly with the various fuel
types and for the life of the car. Unforeseen corrosion can easily contaminate
the fuel delivery system and cause costly repairs.
Manufacturing costs for either tank material seem conflicting, depending on
the source. Nevertheless, due to the invested capital of OEMs on stamping,
welding, and assembly equipment for metal tanks, their cost structure indicates
a lower cost per piece on steel tanks versus plastic ones, with the latter
usually being outsourced (except for some Ford models).
Plastic tanks are formed by blowing a thick continuous tube of HDPE within a
mold that determines the final shape of the virtually seamless part, which
could include the filler neck. The blowing molds are cast from aluminum and
cost considerably less than machined steel dies used to stamp steel tanks. In
general, four or more molds are integrated into one rotary style blow-molding
machine to achieve the desired productivity (i.e., one station blows while the
other one cools). Typically, the OEMs outsource the plastic tanks to various
suppliers who bid for the business. The plastic tank manufacturer also has to
either chlorinate or fluorinate the plastic to retard permeation, and both
processes can be highly toxic if mishandled. This represents additional OSHA
requirements, which add to the cost of the tank.
Plastic tanks have the ability to meet packaging constraints with
complex shapes, and design engineers have greater flexibility in the car design
and styling without having to worry about fitting the gas tank. The plastic
tank could virtually be made to fit whatever cavities are left by the design.
Other attachments to and within the plastic tank require gasketed mechanical
joints. However, plastic swells with constant exposure to organic liquids and
vapor, thus making the joints very critical in the event of repairing the tank
hardware.
| TANK MATERIALS AND ORIGINAL EQUIPMENT MANUFACTURERS |
Chrysler
Chrysler made the decision to outsource plastic tanks in 1988 and, given
recent developments in multilayer HDPE technology, they remain committed to
plastic tanks. The listed advantages of plastic over steel according to
Chrysler include lack of corrosion concerns, easier packaging, ability to
design thickness where needed, and lower weight. Whether all of these
attributes offset the higher unit cost of plastic tanks remains uncertain.
Their plans indicate that all of their tanks will be made of plastic by the end
of 1996. Eventually, all plastic HDPE or sulfonated HDPE tanks will have to be
switched to the more expensive and slower-to-process coextruded ethylene-vinyl
alcohol/HDPE layered tanks—a must for the tanks to pass stricter permeability
standards. This mandatory switch represents an opportunity for steel tanks,
which will be more cost competitive against the coextruded plastic tanks.
Ford
Ford's timetable calls for a switch to Zn-Ni-coated steel from terne-coated
steel tanks in all their models by 1997. Ford is currently using Zn-Ni on the
Bronco, Grand Marquis, Explorer, the new F-150 (PN96), and the new Taurus
(DN101). The switch to Zn-Ni will also include the plastic tank in the current
Escort/Tracer but other models with plastic tanks will remain the same (e.g.,
Ranger, Windstar, Aerostar, and Contour/Mystique).
General Motors
General Motors has an ongoing corrosion test program that includes two-side
painted Zn-Ni, hot-dipped galvanneal, and hot-dipped tin. Zn-Ni-coated tanks
are currently used on the J (Cavalier/Sunfire) and N platforms (Grand
Am/Skylark) and the new fullsize van (GMT 600) launched in November 1995. The
company's current plans include a conversion of all of their current terne fuel
tanks to either Zn-Ni or galvanneal fuel tanks by 2000. |
The average gas tank for a compact automobile (e.g., Nissan Altima) can boast
weight savings of up to 30% versus a similar steel tank.14 However,
Cadillac claims that although their plastic tanks allow design flexibility with
increased safety, they do not achieve any weight savings over steel
tanks.15 These two examples seem to contradict the general view that
plastic's weight advantage increases with the size of the tank. On the other
hand, the new permeability requirement is expected to diminish the weight
advantage of plastics.
One critical part of the performance criteria of the tank is its
ability to meet crash requirements. Generally, plastic tanks are considered
safer in crashes because they are seamless and, thus, not prone to failures in
the vulnerable seam areas. They are not a source of sparks. Also, plastic tanks
deform and have some ability to rebound back to shape. When steel tanks absorb
energy and deform, the pressure within the tank increases as the volume
decreases. This makes them vulnerable at welded or clamped areas where failure
can potentially occur.
The thermal properties of the chosen material are also an issue, especially
due to the proliferation of injector fuel delivery systems, where a portion of
the unused fuel delivered by the gas pump is returned to the gas tank at
"engine-hot" temperatures. At the same time, the tank must withstand extreme
temperatures in North America from -40°C to 79°C in-tank temperatures.
The 79°C temperature not only exceeds the boiling point of the alcohol
fuels, but also creates sagging problems for plastic (especially under the
weight of a filled tank) while the extreme cold introduces potential cracking
problems. As a result, OEMs resort to heavier gauge plastic, negating at least
some of the weight advantage, and must also use support brackets and special
shields against the heat of local sources like an inferior or perforated
muffler or tailpipe. High ambient temperatures underneath the car remain a
consideration.
Plastic acts as an insulator to retard heat transfer to the fuel when compared
to a steel tank. In the case of an under-car fire, plastic tanks will retard
the rise in fuel temperature, but they will soften, sag, and eventually release
the fuel. A steel tank does not sag in a fire; however, the fuel temperature
may rise rapidly, perhaps resulting in over pressurization and release of fuel
through a mechanical fitting. The American Iron and Steel Institute
reports16 that a series of more than 75 tests undertaken by the
National Fire Prevention Research Foundation and Factory Mutual Research
Corporation indicated that plastic containers storing flammable or combustible
liquids in general purpose warehouses fail abruptly when exposed to a small
fire. This failure results in a rapidly developing spill fire that overpowers
conventional sprinkler systems. The same tests conducted with flammable and
combustible liquids stored in steel containers resulted in no spill fire, no
excessive temperatures, no content involvement, and no significant loss of
visibility due to smoke. The fires involving the steel containers extinguished
themselves. These findings have led to a return to steel containers from
plastics for safety and fire insurance cost reasons. It is not known if tests
have been conducted by OEMs to compare the performance of steel and plastic
tanks in under-car fire situations.
Corrosion is a well-known concern on both the inside and outside
surfaces of tanks. The outside surfaces and supporting structure are exposed to
road chemicals, salt, mud, and gravel. The corrosion issue is critical with
zinc-coated products that replace terne plate because of their sacrificial
nature, which puts an even higher demand on the quality of the barrier film for
both inside and outside surfaces. In contrast, the HDPE gas tanks are inert to
the corrosive environments inside and outside the tank.
| Table III. Competitive Materials Analysis for Gas Tanks
|
|---|
Performance Attributes
Recyclability, safety, corrosion resistance in methanol fuels, and weight
|
Manufacturing Issues
Cost, formability/shape flexibility, weldability
|
Steel
- Terne-Coated Steel
- Advantages: Low cost at high volumes, recyclable, materials cost, and permeability
- Disadvantages: Shape flexibility, ineffective corrosion protection from methanol fuel, lead-containing coating
- Electrocoated Zn-Ni and Galvanneal
- Advantages: Low cost at high volumes, recyclable, effective inside and outside corrosion protection, material cost, and permeability
- Disadvantages: Weldability and shape flexibility
- Hot-Dipped Tin
- Advantages: Low cost at high volumes, recyclable, effective inside and outside corrosion protection, material cost, permeability, and weldability
- Disadvantage: Shape flexibility
- Stainless Steel
- Advantages: Corrosion, recycable, and permeability
- Disadvantages: Cost at all volumes, formability, and joinability
|
Plastics
- HDPE
- Advantages: Shape flexibility, low tooling costs at low volumes, weight, and corrosion resistance
- Disadvantages: High tooling costs at high volumes, high material cost, permeability, and recyclability
- Multilayer and Barrier HDPE
- Advantages: Shape flexibility, low tooling costs at low volumes, weight, corrosion resistance, and permeability
- Disadvantages: Higher tooling costs at high volumes, higher material cost, and harder to recycle
|
The Resource Conservation and Recovery Act discourages the use of
materials that cannot be recycled and might end up in landfills.17
As a result, automotive-design engineers must not only meet customer, design,
styling, cost, weight, and regulatory needs but also environmental criteria.
All materials suppliers must show that their product is not only lighter and
cost effective but also recyclable. In this respect, plastics must work the
hardest to show that they are recyclable and have the ability to be recovered
in vehicle disassembly in a cost-effective manner. To accomplish these
objectives, automotive designers must develop prototypes that can be
disassembled easily into the various material groups that have a recycling
infrastructure.
Despite progress in recycling, the proliferation of plastics in automotive
applications faces some hurdles.
- The absence of a plastics recycling infrastructure. The infrastructure for
recovering and recycling the ferrous content of cars is well
established—70-80% of a typical passenger car is recoverable steel and iron.
- The molding process for plastic fuel tanks. As is the case for other
ap-plications, this process results in roughly 30% of plastic material ending
as industrial waste.18
- The necessary sorting of the various plastics types since mixing
types can ruin the batch. This is not a problem with steel—the scrap industry
recycles 10.8 million tonnes per year of shredded automotive iron and steel,
which is used to make new steel products.19
- The lack of technology that dismantlers can use to quickly
collect various plastics. The current infrastructure of automobile dismantlers,
shredders, and scrap-metal processors is steel dominated and relies on magnetic
separation and inexpensive shredding equipment for efficient and low-cost
processing. In the case of terne gas tanks, the units are removed from the car,
flattened, baled, and sent either to special operators that can reclaim the
lead or to the steel mill.
- Cost. Recycled plastics are not cost competitive with virgin
plastics.20
While the recycling of HDPE gas tanks is easier to tackle in terms
of dismantling and avoiding type mixing, these tanks will not meet the new evaporative emissions standards. The barrier-type and multilayer tanks that
will meet such standards can pose a bigger challenge to recycle in a
cost-effective manner.
Table III summarizes the advantages and disadvantages of steel and plastic
with respect to the performance and manufacturing objectives for gas-tank
applications. The comparative analysis of the performance attributes of the
various plastic and steel alternatives indicates that steel products still
represent a cost-effective material that meets all the required performance
criteria of gasoline tanks. A description of current OEM tank activity is
described in the sidebar.
1. Timothy T. Maxwell and Jesse C. Jones, Alternative Fuels: Emissions, Economics and Performance (Warrendale, PA: Society of Automotive Engineers, 1995), pp. 29-42.
2. Robert Q. Riley, Alternative Cars in the 21st Century (Warrendale, PA: Society of Automotive Engineers, 1994), pp. 173-176.
3. Bundy International, "Fuel Supply Systems for a Healthier Environment", ed. Michael Scarlett, Automotive Technology International '94, pp. 37-40.
4. "Plastic Bounces Back in Fuel Tanks," Automotive News (January 30, 1995).
5. Delphi VII Forecast and Analysis of the North American Automotive Industry (Ann Arbor, MI: Office for the Study of Automotive Transportation, University of Michigan Transportation Research Institute, 1994), pp. 71-75.
6. Automotive Outlook: June 1995, (West Chester, PA: Autofacts, Inc.), pp. 4-1, 5-1.
7. "Canadian Blow Molder Expands to U.S.," Canadian Plastics (February 1993).
8. "Ford's Plastic Plant Eyes $110 Investment in Gas Tanks," Plastic Week (August 10, 1992).
9. "Supplier Profile: Walbro Entering Fuel Tank Market; Manufacturer Looks to Double Sales," Ward's Automotive Reports (August 24, 1992).
10. "Air Products Develops Technology that Helps Plastic Fuel Tank Manufacturers Meet More Stringent Emission Standards," Waste Treatment Technology News (December 1992).
11. "Gas Tank Molder Unfazed by CARB," Plastics World (May 1992).
12. "AeroTec Fuel Tanks Designed for Safety," Plastics News (April 12, 1993).
13. "Cadillac Unveils Shift from Terne to Plastics," American Metal Market (January 27, 1992).
14. "Plastic Gas Tank Field is Filling Up," Plastics News (May 11, 1992).
15. "Allante to Use Plastic Tank," Plastics News (January 13, 1992).
16. Steel, the Safe Solution for Flamable Liquid Storage, CN-06, (Washington, D.C.: AISI, 1990).
17. "Recycling and the Automobile," Automotive Engineering (October 1993), pp. 42-50.
18. Asian Autotech Report, 186, (3) (1994), p. 11.
19. "Steel Recycling Increases in '94," American Metal Market (April 5, 1995), p. 10.
20. "Missing Link; Lack of Infrastructure Poses Huge Hurdle to Plans to Recycle Auto Plastics," Automotive News (Supplier Spotlight) (March 14, 1994).
ABOUT THE AUTHOR
Peter J. Alvarado earned his M.B.A. in operations research and his M.Sc. in materials and metallurgical engineering at the University of Pittsburgh in 1981. He is currently market development coordinator at U.S. Steel.
For more information, contact P.J. Alvarado, U.S. Steel, 201 W. Big Beaver Road, Suite 1400, Troy, Michigan 48084-4171; (810) 740-3484; fax (810) 740-5179; e-mail ALVASTEEL@AOL.COM.
Copyright held by The Minerals, Metals & Materials Society, 1996
Direct questions about this or any other JOM page to jom@tms.org.


