 |
48 (11) (1996), pp. 26-28. JOM is a publication of The Minerals, Metals & Materials Society |
|---|
 |
48 (11) (1996), pp. 26-28. JOM is a publication of The Minerals, Metals & Materials Society |
|---|
| CONTENTS |
|---|
Author's Note: As you read this article, consider, for a moment, the possible reaction of a U.S. Occupational Safety and Health Administration inspector if one were to visit the facilities described.
Figure 1 is not taken from a wall painting in a 3rd millennium b.c. Egyptian tomb in the Valley of the Kings. Nor is it a recent archaeometallurgical discovery; but its compositional elements indicate that it could be. In reality, it is a photo of a late 20th century operating aluminum foundry in Ghana, West Africa.
This operating aluminum foundry represents the ingenuity that people can apply when confronted with limited resources and the incentive associated with the need to provide a livelihood for themselves and those dependent upon them. Here, the net result is the making of a consumer item — specifically, the casting of an aluminum pot. What might appear, at first glance, to be a primitive operation is, in fact, an ongoing entrepreneurial business that is making a product for which a local market exists (Figure 2).
 |
| Figure 2. Cast aluminum pots in the market place. |
The cast aluminum pot serves several functions. Its versatility accounts for its popularity in the many local village households where it serves as the primary family cooking utensil. Normally it is suspended from a tripod by its two integral external ears. The tripod itself is typically constructed of 75 mm diameter tree limbs about two meters long. The pot hangs about 300400 mm above the ground level. Small branches, wood scraps, and, sometimes, charcoal provide heat from an open fire built on the ground under it.
But it is more than a cooking pot. Its rounded-bottom shape provides good thermal efficiency. It is also used as the mortar element for the making of the regional staple food called fu-fu — a mixture of yams, casabas, and sometimes grains, which are beaten to a pulpy consistency in the pot by using wooden poles as the pestle element (Figure 3). Water is subsequently added, and cooking proceeds. Other condiments may be added to enhance taste and flavor. The versatile aluminum pot is also used for the various washing requirements of the household.
 |
| Figure 3.Making fu-fu, the regional food staple, using the mortar (pot) and pestle (pole) method. |
Prior to the arrival of an aluminum reduction plant operation in the country, local residents used pots made of cast iron, which came from a distant place. It did not take long for the local entrepreneurs to determine that a similar pot made of aluminum was much lighter, had much better heat-transfer characteristics, and provided an improved and uniform thermal dispersion into the substance being cooked. These attributes are an important economic consideration where fuel is both scarce and expensive.
It is somewhat ironic that the formerly utilized and displaced cast iron pots are used as the patterns for the sand molds made for casting the aluminum. Such a cast iron pot used for pattern purposes is visible in Figure 4, which shows the sand mold being prepared.
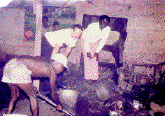 |
| Figure 4. Preparing the sand mold using a cast iron pot as the pattern. The author appears at the left in the picture. |
This aluminum foundry enterprise demonstrates technical ingenuity applied to the limited resources available. The raw material aluminum is the major cost of component of the operation, and its procurement from various sources and different physical conditions is an ever-present economic concern.
The melting furnace at the foundry consists of a hole dug in the ground, approximating 1.5 meters in diameter and one meter deep. The dirt walls and floor are lined with a single layer of loosely stacked used refractory bricks salvaged from the disposal area of a nearby industrial plant.
A cast-iron caldron with a flaring, tapered sidewall (with a top diameter of 750 mm and depth of 500 mm) serves as the melting crucible. The space between the refractory floor and side wall and the crucible provides access for adding the fuel and serves as the combustion chamber. Melting was accomplished by thermal conduction through the cast-iron crucible. Figure 5 is a schematic view of the furnace.
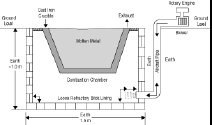 |
| Figure 5. A schematic of the melting furnace. |
Adding to the uniqueness of the foundry operation is its locale; liquid fuel or gas is either prohibitively expensive or unavailable. Solid fuel such as charcoal, coal, or wood is scarce and used only for domestic purposes. Under these conditions, the energy source for an aluminum melting operation manifests the adeptness at transforming a waste nuisance into a valuable resource. The fuel consists of discarded automobile tires, cut up to lengths of 250350 mm. These are fed into the chamber space between the refractory wall and the crucible.
Of course, the burning of the tire fragments by themselves with a natural draught would not provide the intense 650700°C temperatures necessary for melting aluminum. Accelerated combustion is achieved by an improvised tuyere system that provides the required air blast to accelerate the combustion process. The 70 mm diameter air blast pipe enters the ground and extends to the floor level of the furnace.
The air blast had to be supplied by a supplementary source. This consisted of the adaptation of a drum-type blower fan taken from a discarded truck heater system. It was driven at high speed by a gasoline engine of a rotary lawn mower.
Considerable smoke emanates from the burning tire fuel when the furnace is under full fire (Figure 6). While this does not seem to bother the workers, many contemporary cast-shop practitioners may abhor these heavy smoke emissions. However, such a judgement might well be tempered with the consideration that an environmental blight is being alleviated. Specifically, useless, discarded automobile tires strewn about the countryside are collected and disposed of in a productive manner while the effective disposal of discarded vehicle tires continues to be an environmental source of woe in all parts of the world.
 |
| Figure 6. Smoke emanating from the furnace under full-fire conditions. The open-sided lean-to provides ventilation. |
Those who loathe smoke in the work area should be reminded that during the 1940s, most U.S. aluminum melting operations were coal fired. The only provision for smoke, particulate, or fume controls was the post-combustion smokestack. Invariably, the coal used was bituminous, which is notorious for its smoke and volatile gas emissions. The mechanical stokers, with forced air blowers, required two attendants for the fire box and the pulling and shoveling of the ashes and clinkers. Smoke and dust were part of the operation. Thus, it will take third world countries some time to comply with standards beyond their level of current capability.
 |
| Figure 7. Ending the relay pouring. The haziness in the photograph is caused by the gaseous working conditions. |
A somewhat amazing aspect of the foundry operation is the absence of any personal safety equipment for the workers, who exhibit no fear of the molten metal they are handling — a pair of shorts is the only article of clothing in evidence (Figure 8). There is no eye protection; face shields; caps; gloves; or body, arm, and leg coverings. Bare, unprotected feet provided the means of locomotion within the foundry when transferring molten metal from the furnace to the mold area, a distance of 5 m.
The actual metal transfer and casting is accomplished by a three-person relay. Each worker holds a ladle, containing about 4 kg of metal, with tongs that are about 1.5 m in length. Molten metal is dipped from the furnace (Figure 8) and poured into the mold. The sequential execution is a marvel to behold. Every worker seems to know and anticipate every move of their coworkers. There is no wasted motion. Undoubtedly, this teamwork familiarity and dexterity preclude accidents from occuring during operations.
 |
| Figure 8. A close-up view of the top of the furnace showing metal being ladled. The railroad rail is used as a weight to hold the sheet iron cover in place during firing. |
The molten metal is judged to be ready for casting by a visual stirring appraisal; there is no fluxing capability. Skim is removed from the metal surface prior to ladling and broken into small pieces while hot. Subsequently, the skim pile is manually picked over for any identifiable aluminum pieces, which are remelted.
Breaking open the mold after the pour is an unpredictable procedure. A 50 percent rejection rate is considered acceptable and is primarily associated with all of the known sand-casting defects (e.g., laps, seams, holes, cracks, inclusions, and underfill). Nondestructive testing consists of a light hammer tap; the right "ring" signifies no cracks. Visual inspection discloses surface defects. Lifting the pot upside down toward the sky reveals the tiniest holes (Figure 9). Rejected pots are immediately recycled back to the melting furnace. The alloy composition is not controlled and consists of what is primarily available in the area, approximating 99.50% aluminum (1050 alloy).
Obviously there is an abundance of easily identifiable enhancement opportunities at the foundry. But, when the restraint of capital expenditure availability to acquire equipment and supplies from other parts of the world is considered, the opportunities evaporate. Some items such as fluxing materials, a pyrometer, fluidity tester, filter screens, a short trough, a tundish to the mold area to allow a continuous pour, and even a sheet-metal chimney hood to exhaust smoke and fumes would be beneficial. Safety equipment of all types would help. But the reality of the existing circumstances indicate that implementation of these improvements could be problematic.
 |
| Figure 9. A cast aluminum pot in the as-cast condition. It was displayed at the 1996 TMS Annual Meeting Exhibition. |
One suggestion made by the author was to add 57% silicon to improve the fluidity of the molten metal and more than double the temperature range between the liquidus and the solidus. This should facilitate mold filling and reduce the reject rate. But this brought about the question of where the workers could obtain the silicon.
This seemingly anachronistic operation may not provide the substance for an enlightening or humbling experience in the technical arena, but it most certainly does in the realm of human occupational endeavors. This is especially relevant during these periods of reduced earnings, redundancy, job elimination, curtailment of operations, and early retirements. The real question is "are we doing the most (our best) with the resources available to us?" In such a context, this foundry provides an inspiration for facing the adversities created by limited resources and subduing them through ingenuity.
For more information, contact G.J. Binczewski at SCSystems, P.O. Box 6154, Moraga, California 94570; (510) 284-5007; fax (510) 376-5592.
Direct questions about this or any other JOM page to jom@tms.org.
| Search | TMS Document Center | Subscriptions | Other Hypertext Articles | JOM | TMS OnLine |
|---|