 | 49 (1) (1997), pp. 14-17. JOM is a publication of The Minerals, Metals & Materials Society |
|---|
 | 49 (1) (1997), pp. 14-17. JOM is a publication of The Minerals, Metals & Materials Society |
|---|
| CONTENTS |
|---|
This article demonstrates how the fields of science and conservation have collaborated at the National Gallery of Art. Although seemingly unlikely bedfellows, the two disciplines are, in fact, complementary and caution against underestimating the importance of arguably incongruous combinations.
That science and art are unlikely partners is rightfully assumed by those who are proficient at one, but live in great trepidation of the other. Indeed, few can presume to reach the level of excellence achieved by Leonardo da Vinci in both disciplines. Yet, as technology advances, the interdependency of the two professions becomes ever stronger. In the Conservation Division of the National Gallery of Art in Washington, D.C., this unlikely partnership exists daily. Collaboration among scientist, conservator, and curator is fundamental to the common goal of preserving the nation's collections. Whether the objective is to determine the physical condition of a work of art, characterize the materials and methods of a particular artist, or identify a forgery, conservation scientists and conservators use a variety of industrial tools to examine, diagnose, and treat the museum's collections. In this article, case studies that illustrate how modern instrumentation is tailored to meet the needs of the museum world.
When National Gallery curators questioned the attribution and authenticity of a series of small bronze busts of Pope Paul III Farnese (1468-1549), attributed to Guglielmo della Porta (c. 1516-1577) (Figure 1), conservation scientists and conservators were consulted on the viability of such a group. No documentation on the busts prior to 1936 existed, although the sculptures ostensibly dated from an era well known for its meticulous record keeping. Furthermore, no precedent for a group of small-scale portrait busts was known to have existed in the Renaissance.
 |
| Figure 1. A series of seven small-scale busts of Pope Paul III Farnese, formerly attributed to Guglielmo della Porta. |
In the lost-wax technique, cire perdue, a wax model (built over a solid core if the final sculpture is to be hollow) is encased in mold material. Molds can be made of any refractory material—a material that will withstand high temperatures without changing shape. Traditionally, plaster was used. The mold is heated and the wax is melted out so that the shape of the model is retained between the walls of the mold and the inner core. Molten metal is poured into the mold and assumes the shape left by the wax model. Finally, the mold is broken open, and finishing touches are made to the metal sculpture.
What became apparent in the first step of the technical examination of the busts was not that the casting method chosen for these bronzes was atypical of Renaissance sculptures, but that the approach to the fabrication of the sculptures was quite similar within the group. Remnants from the casting process such as small holes left by the core pins or excess metal left along the truncation of the bust from the sprues or runners appeared consistent on the sculptures. This, in turn, suggested that the busts were made by the same individual or workshop that had achieved a working method that was capable of yielding consistent results.
 |
| Figure 2. Bacchus and a Faun, a 16th century bronze sculpture by an anonymous Italian. (National Gallery of Art, Washington, D.C., Andrew W. Mellon Collection.) |
The analyses were performed on a Kevex 0750A energy-dispersive XRF spectrometer equipped with a BaCl2 secondary target, 6 mm collimators, 60 kV anode voltage, and 0.4 mA anode current. Several areas were investigated from each of the seven busts. Kevex XRF Quantex-Ray software, using the EXACT fundamental parameters program provided by Kevex Corporation, was used for to calculate the elemental surface composition. It should be emphasized that this particular technique analyzes only the uppermost surface (up to approximately 100 µm), and bulk values may vary considerably.
The XRF analyses were performed using a modified freestanding instrument with the x-ray tube and detector mounted on a steel column that allowed for vertical and horizontal movement of the spectrometer. The art work was placed in front of the XRF spectrometer, where a laser and incandescent lamp were used to focus on the desired sample area. This ingenious adaptation of the instrument allows for the analysis of the sculpture without the necessity of removing a sample.
XRF analyses revealed that all seven of the busts were brasses rather than bronzes whose average compositions ranged from 60-73% copper, 23-36% zinc, 2-3% lead, and 1% tin. To confirm the bulk values, a sample was removed from the interior of the bust and ananlyzed by Janet Snyder of the Freer Gallery of Art, Smithsonian Institution, Washington, D.C., using atomic absorption spectroscopy (AA). The surface was determined to be of virtually the same composition as the bulk values. The copper, zinc, tin, and lead values obtained using AA were within 1% of the XRF values. The elements less than 1% were within 40%.
 |
| Figure 3. Cherubs Playing with a Swan, a 17th century lead fountain by Jean-Baptiste Tubi. (National Gallery of Art, Washington, D.C., Andrew W. Mellon Collection.) |
"When they are glowing, take calamine (zinc carbonate) . . . very finely ground, with coals, and arrange them in each cup about one sixth part full, and fill it quite with the above-mentioned copper, and cover with coals. . . . And when the copper is altogether melted, take a slender iron, long and curved and fixed to a wooden handle, and carefully stir it, that the calamine may be mixed with the copper."3
Generally, the zinc content obtained using this procedure ranged from 22-28%.4 In addition, impurities usually found in Renaissance bronzes curiously were not detected in any of the seven busts examined; a fact that introduced the distinct possibility that an electrolytically refined copper had been used. From the assembled evidence, it became apparent that the busts were most likely 19th or possibly 20th century creations.5
XRF analysis was also used to expose anomalies within the same work of art, as seen in the case of Bacchus and a Faun by an unknown Italian sculptor of the 16th century (Figure 2). At one time, the genitals of Bacchus were modestly hidden behind fig leaves. National Gallery curators believed the fig leaves to be more in keeping with Victorian sensibilities than Renaissance decorum. XRF analyses of both Bacchus and the faun revealed the alloy to be a traditional bronze composed primarily of copper and tin; however, the fig leaves were found to be brass. Although the evidence of vastly differing alloys and impurities was not conclusive enough to condemn the fig leaves entirely, the findings lent credence to the curator's hypothesis that they were later additions. Subsequently, the fig leaves were removed.
 a a |
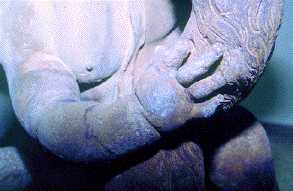 b b |
| Figure 4. A cherub's hand on the lead fountain Cherubs Playing with a Swan: (a) before treatment showing lead carbonate and (b) after treatment. Note the modeling on the swan's neck that was previously not visible. (National Gallery of Art, Washington, D.C., Andrew W. Mellon Collection.) |
At the National Gallery of Art, a conventional Gandolfi camera and film are used to perform XRD analysis. Unfortunately, a goniometer cannot be used, as samples taken from works of art are extremely small. With the aid of a stereomicroscope, a few grains of the corrosion are removed with a tungsten needle or a scalpel and placed onto the tip of a glass fiber. If desired, the sample can be removed from the fiber for further examination.
XRD revealed the obscuring film to be lead carbonate.6 The analyses were performed on a Philips x-ray generator 3100 equipped with a copper target and nickel filter to provide Cu K alpha radiation, 45 kV anode voltage, and 25 mA current. Since lead carbonate is stable, and its removal would disrupt the equilibrium of the fountain's surface, a solution had to be reached to minimize the unsightly appearance created by the lead carbonate, while leaving enough of a film to retain the relative surface stability.
Conservators turned to dentistry to resolve the dilemma. They selected a cavitron, an ultrasonic dental unit, as the cleaning tool of choice. Ultrasonic vibration causes the formation of tiny bubbles in the water pumped through the cavitron that ultimately dissolves superficial deposits without harming the substrate. However, not just any water could be used with the cavitron. Unfiltered water running through the fountain for many years had, after all, been the source of the damage. For cleaning the fountain with the cavitron, water filtered through a deionizing column was used. The cavitron reduced the accumulation of surface deposits and corrosion, producing a more cohesive appearance while revealing remnants of gilding and polychrome.
 |
| Figure 5. The Thinker, a late nineteenth century bronze sculpture by Auguste Rodin. (National Gallery of Art, Washington, D.C., gift of Mrs. John Simpson.) |
Science and conservation have recently begun to add a technical dimension to catalogues of the National Gallery collections. Among one of the many systematic cataloguing projects is a volume dedicated to 19th century French sculpture. In this volume, each sculpture entry covers a range of technical information including materials employed, fabrication methods, and condition. X-radiography, one of the most useful tools in these examinations, is performed using an industrial x-ray unit with a 350 kV capability. It is housed in a custom-designed room with walls of steel and lead and a floor reinforced with two feet of concrete to support the weight of the walls.
Perhaps the most well known sculpture included in the volume is The Thinker by Auguste Rodin (Figure 5). Conceived originally as part of the grand sculptural composition The Gates of Hell, commissioned by the French government in 1880, Rodin modeled his brooding Thinker for
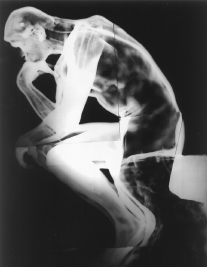 |
| Figure 6. An x-radiograph revealing a lead counterweight in the base of The Thinker and coiling iron armatures within the sculpture. (National Gallery of Art, Washington, D.C., gift of Mrs. John Simpson.) |
In the x-radiograph of the Thinker (Figure 6), the lead anchor is visible as a white mass at the back of the base. Additional information gleaned from x-radiography is that iron armatures remain inside the sculpture. Seen in the x-radiograph as curling gray forms, the armatures were used to hold internal core material in place during casting. (The sculpture is already extremely heavy because of the lead counterweight; it would have been impossibly heavy and considerably more expensive to manufacture had it been cast as solid bronze.) As the statue is displayed in a climate-controlled indoor environment at the National Gallery, and there is no evidence of rust on its surface, conservators did not believe it was necessary to remove the armatures within the piece. Sculptures of The Thinker exhibited outdoors, however, often have the iron armatures removed because, in an uncontrolled outdoor environment, iron is conducive to the formation of rust and, as a result, the degradation of the sculpture occurs.
The authors are grateful to Katherine Holbrow, Mellon Fellow, at the Objects Conservation Department and the National Gallery of Art for sharing the information on the conservation of the fountain.
1. B. Cellini, The Treatises of Benvenuto Cellini on Goldsmithing and Sculpture (1568), ed. C.R. Ashbee (London: Publisher, 1898).
2. R.F. Tylecote, A History of Metallurgy (London: Publisher, 1976), p. 96.
3. Theophilus, An Essay Upon Various Arts in Three Books by Theophilus (11th century), trans. Robert Hendrie (London: Publisher, 1847), p. 307.
4. O. Werner, "Uber das Vorkommen von Zink und Messing im Alertum und im Mittelalter," Erzmetal, 23 (1970), pp. 259-269.
5. D. Barbour and L.D. Glinsman, "An Investigation of Renaissance Casting Practices as a Means for Identifying Forgeries," Conservation Research, Monograph Series II (1993), pp. 14-29.
6. JCPDS, Powder Diffraction File Search Manual (Swarthmore, PA: Publisher, 1982).
ABOUT THE AUTHORS
L. Glinsman earned her B.S. in chemistry from the College of St. Rose in 1982. She is currently a conservation scientist at the National Gallery of Art.
D. Barbour earned her M.A. in art history and certificate in conservation from New York University in 1987. She is currently an objects conservator at the National Gallery of Art.
For more information, contact L. Glinsman, National Gallery of Art, Conservation Department, Washington, D.C., 20565; (202) 842-6217; fax (202) 842-6886.
Direct questions about this or any other JOM page to jom@tms.org.
| Search | TMS Document Center | Subscriptions | Other Hypertext Articles | JOM | TMS OnLine |
|---|